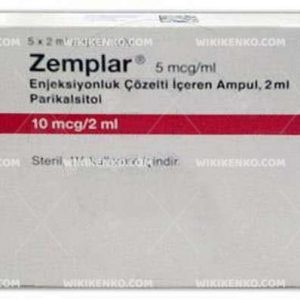
Description
Zemplar injection is a clear, colorless, and particle-free aqueous solution containing the active ingredient paricalcitol, a synthetic vitamin D analog. Each milliliter of the solution contains 5 micrograms of paricalcitol, along with excipients such as ethanol (20% v/v) and propylene glycol (30% v/v).
How Does Zemplar Injection Work?
Paricalcitol, the active ingredient in Zemplar, is a selective vitamin D receptor (VDR) activator. It works by inhibiting parathyroid proliferation, effectively reducing elevated parathyroid hormone (PTH) levels associated with secondary hyperparathyroidism. Unlike traditional vitamin D therapies, paricalcitol has a minimal impact on calcium and phosphorus levels, making it a safer alternative for patients with chronic kidney disease.
Additionally, paricalcitol directly affects bone cells, helping to maintain bone volume and improve mineralization surfaces, which is crucial for patients with chronic kidney disease who often experience bone disorders.
Dosage and Administration
The dosage of Zemplar injection is determined by the patient’s age, condition, and response to treatment. Here are the general guidelines:
Adult Dosage:
- Initiate at 0.04 mcg/kg to 0.1 mcg/kg (2.8 mcg to 7 mcg) no more frequently than every other day.
- Adjust the dose to achieve the desired therapeutic range of intact PTH levels and maintain serum calcium within normal limits.
- Monitor serum calcium levels twice weekly and intact PTH levels every 2 to 4 weeks.
Pediatric Dosage:
- Initiate based on baseline intact PTH levels:
- If < 500 pg/mL, start with 0.04 mcg/kg.
- If ≥ 500 pg/mL, start with 0.08 mcg/kg.
- Monitor serum calcium and intact PTH levels similarly to adults.
Zemplar is administered intravenously through a hemodialysis vascular access at any time during dialysis.
Available Forms
Zemplar injection is available in the following forms:
- Single-dose vials:
- 2 mcg/mL
- 5 mcg/mL
- Multiple-dose vial: 10 mcg/2 mL (5 mcg/mL)
Contraindications
Zemplar should not be used in patients with:
- Hypercalcemia (high blood calcium levels)
- Vitamin D toxicity
| Key Information | Details |
|---|---|
| Side Effects | Common side effects may include nausea, vomiting, swelling in hands or feet, dizziness, joint pain, and runny nose. Seek medical attention for severe symptoms or suspected overdose. |
| Dosage | Adult: 0.04-0.1 mcg/kg every other day<br>Pediatric: Based on PTH levels (0.04-0.08 mcg/kg) |
| Administration | Intravenous through hemodialysis access during dialysis |
| Available Forms | Single-dose vials (2 mcg/mL, 5 mcg/mL)<br>Multiple-dose vial (10 mcg/2 mL) |
| Contraindications | Hypercalcemia, vitamin D toxicity |
| Drug Interactions | Major: Allopurinol, amlodipine, aspirin, atorvastatin, carvedilol, and more<br>Moderate: Cholecalciferol, furosemide, lisinopril, omeprazole, prednisone, sevelamer, and more |
Marketing Authorization
Zemplar injection is manufactured by AbbVie Ltd., headquartered in Maidenhead, UK, and has received marketing authorization in various countries, including Austria, Brazil, Ecuador, Estonia, Spain, Hong Kong, Ireland, Israel, Italy, Lithuania, Mexico, Romania, Singapore, Turkey, the United Kingdom, the United States, and South Africa.
Conclusion
Zemplar (paricalcitol) injection is a valuable treatment option for patients with chronic kidney disease undergoing dialysis, helping to manage secondary hyperparathyroidism and promote bone health. By selectively activating vitamin D receptors and reducing parathyroid hormone levels, Zemplar offers an effective solution with minimal impact on calcium and phosphorus levels.
However, it is crucial to follow your healthcare provider’s instructions, monitor your response to treatment, and report any adverse effects promptly.









Reviews
There are no reviews yet.