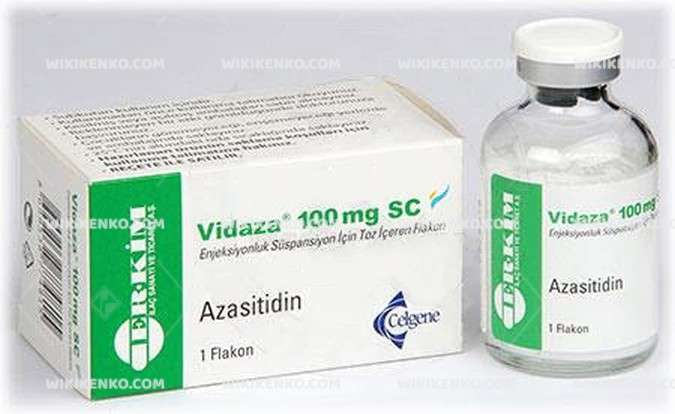
Vidaza Sc Injection Suspension Icin Powder Iceren Vial
Azacitidine is marketed under the trade name Vidaza. It is a chemical analog of cytidine, a nucleoside in DNA and RNA that is used in the treatment of myelodysplastic syndrome. 5-Azacitidine is the other name of this medication. Azacitidine is called “antineoplastic” or “cytotoxic” medication that is classified as an “antimetabolite” and a “demethylation” agent.
| Dosage form | |
|---|---|
| Pack size | |
| Potency | 100 Mg |
| Manufacturer | |
| Origin | |
| Generic Name (Ingredient) | Each Vial Contains 100 Mg Of Azacitidine. The Suspension Obtained After The Preparation Contains 25 Mg Of Azacitidine Per Ml. |
Assuming your emergency circumstances for this product, visit Urgent Quotation page. Besides, for any pharmaceutical questions, please ask us in the comments section.
Description
History
In 2004: Vidaza was approved by the FDA for the treatment of Myelodysplastic Syndromes (MDS).
In 2007: Vidaza NDA Supplement was approved by the FDA for IV Administration.
In 2008: It receives expanded FDA approval to include overall survival in Higher-Risk MDS.
Medical use
This anti-cancer chemotherapy is indicated for the treatment of
- myelodysplastic syndrome (MDS)
- Chronic myelomonocytic leukemia (CMML)
Mechanism of Action
It is believed that Azacitidine have antineoplastic activity via two mechanisms; that means its anticancer effects are twofold
- At low dise: by inhibiting of DNA methytransferase and causing hypomethylation of DNA, normal function to the tumor suppressor genes and control over cell growth are restored.
- At high dose: Azacitidine is a type of chemotherapy called antimetabolites. When the cells incorporate antimetabolites into the cellular metabolism, a direct cytotoxic effect is caused that result in cell death.
Vidaza Market
Vidaza was approved in the USA in May 2004 for myelodysplastic syndromes, a disease of the bone marrow which may progress to leukaemia. Sales of Vidaza for the third quarter were up 15.6% to $42.3 million and Pharmion recently announced new Phase III data which demonstrated that patients receiving the drug had a two-year survival rate of 50.8% (or 24.4 months) versus 26.2% (or 15 months) for those on conventional care regimens.
Pharmion Corporation (NASDAQ: PHRM) today reported financial results for its quarter and year ended December 31, 2007. For the year, net sales totaled $267.3 million, compared to net sales of $238.6 million in 2006. Worldwide sales of Vidaza® (azacitidine for injection) totaled $165.3 million for 2007, compared to $142.2 million for 2006. In the U.S., sales of Vidaza totaled $131.4 million in 2007, compared to $130.6 million in 2006.
“We are very pleased with our financial results for 2007, and very proud of the major milestones we accomplished during the year,” said Patrick J. Mahaffy, Pharmion’s president and CEO. “We were very excited about the results of the Vidaza overall survival study announced in August, the increasing uptake of Vidaza for European named patient and compassionate use that fueled Vidaza sales growth during the year, as well as significant progress in clinical research across our product portfolio, including the initiation of clinical studies for Amrubicin, MGCD0103 and oral Azacitidine.”
Celgene Corp is splashing out $2.9 billion in cash and stock to acquire fellow US firm Pharmion Corp as part of its bid to become “a global leader in the haematology/oncology field”. Under the terms of the agreement, Celgene will acquire Pharmion for $72.00 per share, payable in a combination of cash and stock. The deal represents quite a leap on Pharmion’s closing price on Friday night of $49.28.
Celgene chief executive Sol Barer said the purchase is “an exceptional strategic fit that will expand our role as a leader in haematology and oncology”. He added that “our combined global infrastructure will leverage the therapeutic and commercial potential of Pharmion’s products, particularly Vidaza (azacitidine) which has the potential to become a major global therapy.
Market authorisation for Vidaza in higher-risk MDS will be sought in Europe before the end of the year and analysts believe that Celgene will be able to market its best-selling drug Revlimid (lenalidomide), an analogue of thalidomide, for less sick patients with MDS, then switch patients to Vidaza after they develop resistance.
Panacea Biotec Ltd. announced that it has entered into a tripartite agreement with Natco Pharma Ltd. and Breckenridge Pharmaceutical Inc. for the manufacture and supply of Azacitidine injection for the US market undeBreckenridge’s already-Approved New Drugs Application (ANDA). Azacitidine is a generic equivalent of Vidaza. It is a chemotherapy drug used to treat conditions that affect the blood and the bone marrow, called myelodysplastic syndromes (MDS). It is used when treatment with a stem cell transplant is not suitable. The annual sales of the drug is about US$140 million, as reported by IMS, MAT, Dec 2017. Natco has provided the technology for Azacitidine to Panacea Biotec, which will manufacture and supply the product to Breckenridge in the US to market, sell and distribute. The technology has been provided to Panacea Biotec’s facility located at Baddi, Himachal Pradesh, India. The application (prior approval supplement) for qualifying Panacea Biotec’s site has been filed with the US FDA and approval is expected in due course of time. This collaboration will enable the company to grow its revenues and will also ensure increased capacity utilization of its oncology plant at Baddi.
Use the form below to report an error
Please answer the questions as thoroughly and accurately as possible. Your answers will help us better understand what kind of mistakes happen, why and where they happen, and in the end the purpose is to build a better archive to guide researchers and professionals around the world.
The information on this page is not intended to be a substitute for professional medical advice, diagnosis, or treatment. always seek the advice for your physician or another qualified health provider with any questions you may have regarding a medical condition. Always remember to
- Ask your own doctor for medical advice.
- Names, brands, and dosage may differ between countries.
- When not feeling well, or experiencing side effects always contact your own doctor.
Cyberchondria
The truth is that when we’re sick, or worried about getting sick, the internet won’t help.
According to Wikipedia, cyberchondria is a mental disorder consisting in the desire to independently make a diagnosis based on the symptoms of diseases described on Internet sites.
Why you can't look for symptoms on the Internet
If diagnoses could be made simply from a textbook or an article on a website, we would all be doctors and treat ourselves. Nothing can replace the experience and knowledge of specially trained people. As in any field, in medicine there are unscrupulous specialists, differences of opinion, inaccurate diagnoses and incorrect test results.


Reviews
There are no reviews yet.