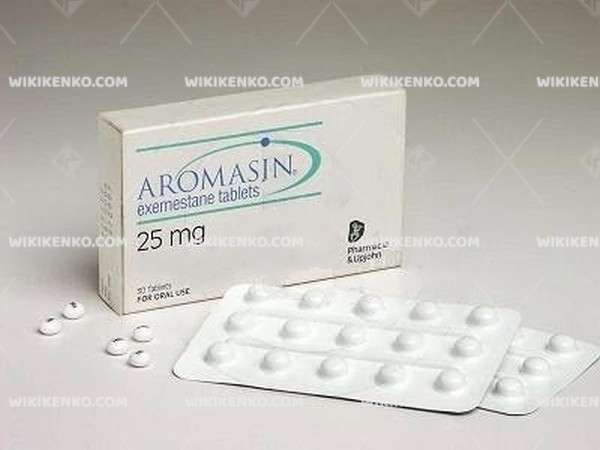
Description
Aromasin Medical Uses
This aromatase inhibitor is indicated for
- the treatment of advanced breast cancer in breast cancer in women who no longer have their menstrual periods, either naturally due to their age (after menopause) or because they have had their ovaries surgically removed.
- risk reduction for invasive breast cancer in postmenopausal women.
Mechanism of action
In premenopausal women, ovaries are the main sources of estrogen but converted estrogen by the aromatase enzyme in the peripheral tissue and some parts of the brain is the source of estrogen in post-menopausal women. Exemestane is acted as an intermediate and binds to the active site of the enzyme. This binding leads to inactivation known as ” suicide inhibition”. This medication is structurally similar to the enzyme targets; si it permanently binds to the enzyme, preventing them from converting androgen into estrogen.
Market Trends of Aromasin
Pfizer Inc, the main medicine wholesaler has received approval from the U.S. Food and Drug Administration (FDA) to market Aromasin® (exemestane tablets). These tablets are used for adjuvant treatment of postmenopausal women with estrogen-receptor positive early breast cancer following two-to-three years of tamoxifen for completion of five consecutive years of adjuvant hormonal therapy.
This approval was based on the Intergroup Exemestane Study (IES). During this study, it was shown patients who switched to Aromasin after two-to-three years of tamoxifen, for a combined total of five years of therapy, had 31 percent more protection from cancer recurrence than those who remained on five years of tamoxifen therapy. This landmark published a study in the New England Journal of Medicine, has shown the superiority of switching to Aromasin rather than remaining on tamoxifen. After releasing the results of this study and their publication, the American Society of Clinical Oncologists and the National Comprehensive Cancer Network updated their guidelines to support the use of a new switch regimen using Aromasin adjuvant treatment.
Dr. Joseph Feczko, a chief medical officer in Pfizer, the main pharmaceutical exporter declares that” Pfizer is pleased to announce this treatment advance. This new indication for Aromasin provides women with a new treatment regimen that can significantly improve their chances of remaining breast cancer-free.” Dr. Stephen Jones, medical director at U.S. Oncology Research in Houston, Texas believes that “This new approval for Aromasin clearly provides physicians and patients a more effective way of protecting against cancer recurrence compared to the current treatment practice of five years of tamoxifen therapy”.
The European Union approved Aromasin for adjuvant treatment; it has been marketed by Pfizer. Aromasin was first approved in the United States in 1999 as a treatment for advanced breast cancer in postmenopausal women whose tumors have stopped responding to tamoxifen. It also received approval for using in Europe, Japan, and South America. It is currently available in more than 50 countries around the world.
Mylan N.V. has announced the U.S. launch of Exemestane Tablets, 25 mg; It is a generic version of the reference listed drug, Pfizer’s Aromasin® Tablets. Mylan got final approval from the U.S. Food and Drug Administration (FDA) for it’s Abbreviated New Drug Application (ANDA) for this product; this new product is indicated in the treatment of certain types of breast cancer in women after menopause. Mylan is the largest supplier of cancer medicines by volume in the U.S., and with the launching of Exemestane Tablets, 25 mg, the company’s robust oncology franchise will be higher. According to the IMS Health report, Exemestane Tablets, 25 mg, had U.S. sales of approximately $100 million for the 12 months ending Jan. 31, 2017. Aromasin price is only $1,158 for a supply of 30 tablets.









Reviews
There are no reviews yet.