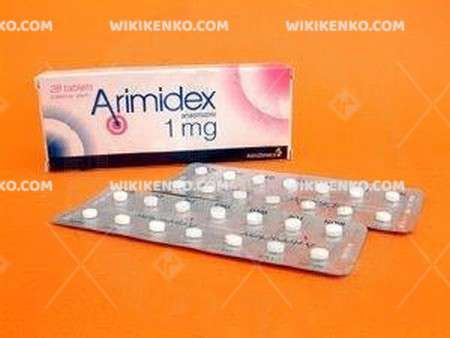
Description
Medical Use
ARIMIDEX is approved
- As an adjuvant treatment for postmenopausal women with hormone receptor-positive early breast cancer
- For the initial treatment of postmenopausal women with hormone receptor-positive or hormone receptor unknown locally advanced or metastatic breast cancer
- For the treatment of postmenopausal women with advanced breast cancer that has progressed following treatment with tamoxifen.
Mechanism of Action
The hormone estrogen stimulated the growth of some breast cancers. In women who have been through the menopause, this hormone is no longer produced by the ovaries. But some estrogen is still made in body fat using a protein enzyme, aromatase. Aromatase inhibitors like Anastrozole stop this enzyme from working so there’s less estrogen in the body. This means it blocks the enzyme aromatase. When there is no estrogen, tumor cells dependent on estrogens grow less. Anastrozole reversibly binds to the aromatase enzyme and blocks the conversion of androgens to estrogens in peripheral (extragonadal) tissues through competitive inhibition.
Arimidex Market
Arimidex (anastrozole) is a new generation Harmon therapy for breast cancer that has been launched in the UK. This is the first world launched the product that is indicated for the palliative treatment of post-menopausal women with advanced breast cancer.
According to data presented at the 4th Nottingham International Breast Cancer Conference in the UK, Arimidex is a highly selective, non-steroidal aromatase inhibitor with improved tolerability and increased convenience compared to other available drugs of this type, such as aminoglutethimide and 4-hydroxy-androstenedione.
Zeneca hopes that Arimidex will eventually be indicated for the adjuvant treatment of early breast cancer. So they are planning an adjuvant study comparing Arimidex to tamoxifen (and possibly to combination treatment) in collaboration with the UK Cancer Research Campaign. According to the data Presented for the first time at the San Antonio Breast Cancer Meeting, Arimidex is not only significantly more effective than tamoxifen in terms of DFS, but also it has a number of important tolerability benefits over the current gold standard include a significant reduction in both the incidence of endometrial cancer and thromboembolic events.
According to the latest ATAC trial results in December 2004, AstraZeneca’s Arimidex (anastrozole) will replace tamoxifen as the gold-standard adjuvant therapy for local and locally advanced breast cancer.
These data reveal an important breakthrough in the management of this increasingly prevalent disease because this is the first time that the established benefits of tamoxifen in early breast cancer have been surpassed by another treatment.
AstraZeneca announced that its Japanese operating company has been able to break out the restriction to the Arimidex 1 mg prescribing information in Japan regarding the use of Arimidex (anastrozole) as adjuvant therapy. They did this by notifying this amendment to the Japanese health authority (MHLW)in 2002.
So Japan has changed to the first world market to promote Arimidex as adjuvant therapy but it is already licensed worldwide for use in advanced disease. Extended license applications have already been submitted to the US FDA and European regulatory authorities but Although Japan is the first market to promote Arimidex for early breast cancer.
Dr. George Blackledge, Vice-President and Medical Director of Oncology at AstraZeneca, a medicine company Welcoming this first launch for Arimidex in early breast cancer and stated that “It is excellent news that we are starting to bring the proven benefits of Arimidex to the many women newly diagnosed with early-stage breast cancer. The results of ATAC are truly groundbreaking for the management of this devastating disease, and we look forward to further license approvals in other markets in the future”.








Reviews
There are no reviews yet.