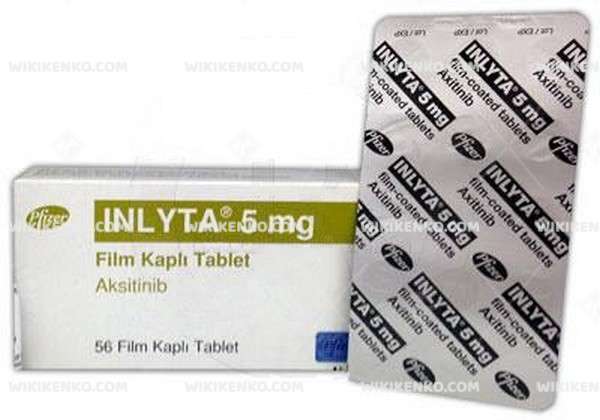
Inlyta Film Coated Tablet 5 Mg
Axitinib is marketed under the trade name Inlyta by Pfizer, one of the main pharmaceutical supply chain for hospitals. It is a targeted therapy, a receptor protein-kinase and also angiogenesis inhibitor. This medication is used as a treatment for advanced renal cell carcinoma and also kidney cancer. The most common Inlyta side effect is diarrhea.
| Dosage form | |
|---|---|
| Pack size | |
| Potency | 5 Mg |
| Manufacturer | |
| Origin | |
| Generic Name (Ingredient) | Each Film-Coated Tablet Contains 5 Mg Aksitinib. |
Assuming your emergency circumstances for this product, visit Urgent Quotation page. Besides, for any pharmaceutical questions, please ask us in the comments section.
Description
Axitinib is marketed under the trade name Inlyta by Pfizer, one of the main pharmaceutical supply chain for hospitals. It is a targeted therapy, a receptor protein-kinase and also angiogenesis inhibitor. T
Axitinib Usage
Axitinib is often used after other cancer medicines have been tried without success. It is indicated for the treatment of
- Advanced kidney cancer
- Advanced renal cell carcinoma
Mechanism of Action
Axitinib is a tyrosine kinase inhibitor; that means this medication is a cancer growth blocker. Tyrosine kinases send growth signals to the cancer cells. This inhibitor blocks tyrosine kinases from acting on the cells. It is also called anti-angiogenesis treatment. During this treatment, axitinib stops forming blood vessels by cancer cells. Cancer cells need these vessels for growing.
Cancer cells divide rapidly and some of the normal cells too. Target therapy is about the differences between cancer cells and normal cells. There are different types of targeted therapy with a lit bit different function, but all of them attach the cancer cells without damaging the normal cells. VEGFR is the abbreviation of vascular endothelial growth factor receptor that is located both on the normal and cancer cells. Axitinib as a targeted therapy binds to this receptor on the cancer cells and blocks the angiogenesis promoting pathway.
Inlyta Market
Inlyta (axitinib) was developed and marketed by the pharma company, Pfizer for treating Advanced Renal Cell Carcinoma (ARCC) and hepatocellular carcinoma. This medication was investigated as a single dose therapy, as well as in combination with chemotherapy. Submission of Inlyta (axitinib) for the treatment of advanced renal cell carcinoma was based on the Phase III AXIS 1032 trial. It was accepted for review by the FDA in June 2011 but Pfizer received final approval for Inlyta from the FDA in January 2012 for the treatment of ARCC. The U.S Food and Drug Administration has approved this drug based on inspection and considering its benefits. The European Medicines Agency accepted to review this drug in June 2011. This review was based on a Phase III trial named AXIS 1032 too. Pfizer received its marketing authorization approval for the treatment of patients suffering from ARCC from the European Medical Authority (EMA) in May 2012. This medication also received marketing approvals for the treatment of ARCC in Australia and the UK in 2012. It also received approval in Switzerland, Japan, Canada, and Korea. Pfizer signed an agreement with SFJ Pharma for the co-development of Inlyta (axitinib) in Asia.
Commercially, the drug demonstrates multiple benefits in particular cases, but situations such as hypertension and arterial and venous thromboembolic events need special attention during diagnosis with axitinib. Generally, the associated benefits enable its viability and it is a proven source of efficient diagnosis of cancer. It is expected that Global Axitinib Market grows at a highly moderate CAGR in a specific period.
Factors such as rising cases of cancers on a global scale drive the Global Axitinib industry. Moreover, changing the lifestyle of consumers and the growing prevalence of chronic illness leading to cell mutation are the main reasons that likely propel the market growth in the future. Besides, rising cases of breast cancers in geriatric women at the global level are likely to be treated with axitinib since it inhibits the growth of breast cancer cells in women. But, it is likely that high costs associated with treatment in such cases and extra precaution needed in patients with hypertension and arterial and venous thromboembolic events impede the market growth in the future. Geographically, the global Axitinib industry spans North America, Latin America, Europe, Asia-Pacific, Middle-East and Africa. In terms of value, North America dominates the global market.
It is expected that extensive research on cancer propels the market growth in the future. Probably European regions follow this trend. It is anticipated that APAC regions grow in the future owing to extensive research and development in cancer and associated fields. The rise in a number of cancer patients and the geriatric population will help the market growth. It is also expected that market in Middle-East and African grow at a moderate CAGR in the future due to rising cases of cancer and growing medical infrastructure.
The key players in the global axitinib industry are Pfizer, Allergan, Alnylam Pharmaceuticals, Amgen, Angstrom Pharmaceuticals, Acceleron Pharma, Biocon, Bionomics, CASI Pharmaceuticals, Celgene Corporation, Enzon Pharmaceuticals, Fujifilm Kyowa Kirin Biologics, Genexine, Hetero Drugs, ImClone Systems, Kyowa Hakko Kirin, Neumedicines, Mabtech, Marsala Biotech, Novartis, Oncobiologics, and Onyx Pharmaceuticals. Axitinib price is around $14,940.03.
his medication is used as a treatment for advanced renal cell carcinoma and also kidney cancer. The most common Inlyta side effect is diarrhea.
Use the form below to report an error
Please answer the questions as thoroughly and accurately as possible. Your answers will help us better understand what kind of mistakes happen, why and where they happen, and in the end the purpose is to build a better archive to guide researchers and professionals around the world.
The information on this page is not intended to be a substitute for professional medical advice, diagnosis, or treatment. always seek the advice for your physician or another qualified health provider with any questions you may have regarding a medical condition. Always remember to
- Ask your own doctor for medical advice.
- Names, brands, and dosage may differ between countries.
- When not feeling well, or experiencing side effects always contact your own doctor.
Cyberchondria
The truth is that when we’re sick, or worried about getting sick, the internet won’t help.
According to Wikipedia, cyberchondria is a mental disorder consisting in the desire to independently make a diagnosis based on the symptoms of diseases described on Internet sites.
Why you can't look for symptoms on the Internet
If diagnoses could be made simply from a textbook or an article on a website, we would all be doctors and treat ourselves. Nothing can replace the experience and knowledge of specially trained people. As in any field, in medicine there are unscrupulous specialists, differences of opinion, inaccurate diagnoses and incorrect test results.


Reviews
There are no reviews yet.